Using well-known mechanisms that determine the epidemiology and stability of invasive antibiotic resistance plasmids in bacteria, we have developed simple and effective tools to remove them from the microflora. This gives us an uniquely positive solution to AMR and restores health to invaded bacterial ecosystems.
Contact
[email protected] & [email protected] [email protected]
Antimicrobial resistance (AMR) curing and protection of the microbiota from AMR re-invasion (1-3). Microbiota protection (2-3) when needed, e.g. before hospital admission or overseas travel.
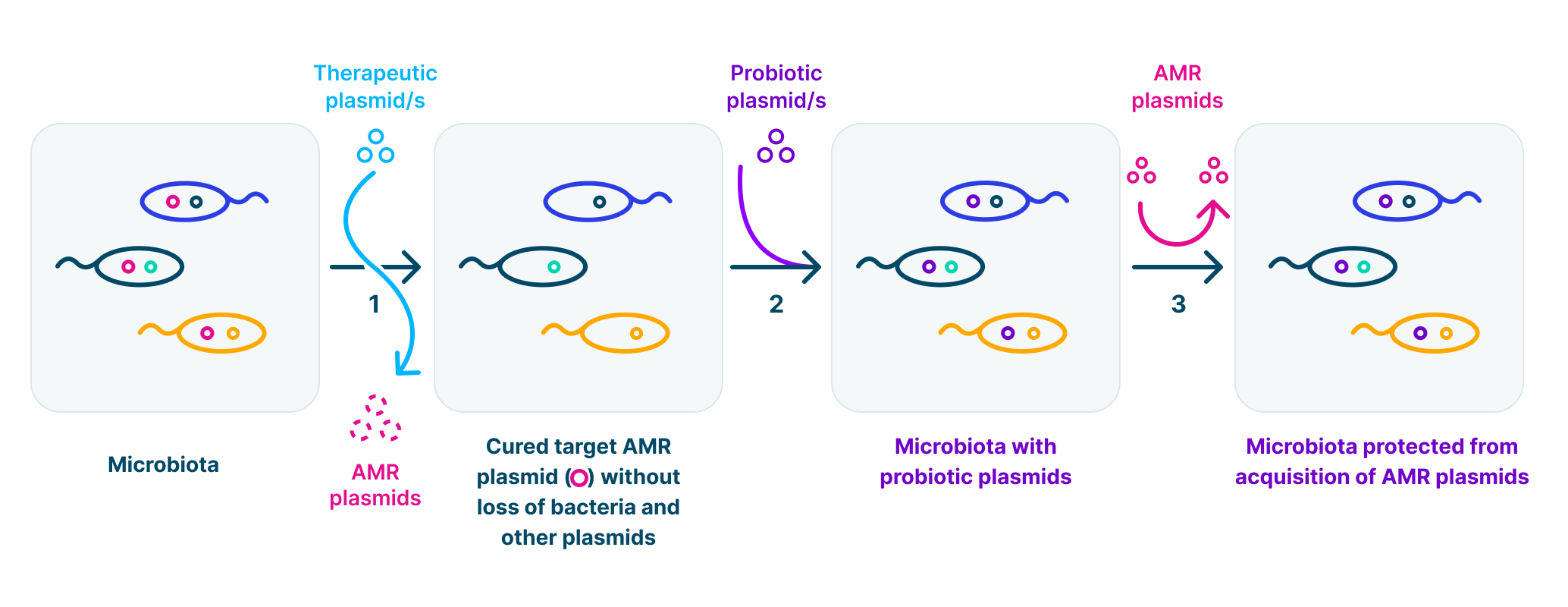
We construct plasmids to treat antibiotic resistance in bacteria. Our genetically modified (GM) therapeutic plasmids can be used to cure antibiotic resistance plasmids from bacteria in humans, animals and environmental ecosystems. Our GM-probiotic plasmids protect bacteria from acquisition of antibiotic resistance plasmids.
This procedure cures antibiotic resistance without killing of host bacterial populations. This preserves the microbiota and prevents ecological damage.
We expect to conduct first-in-human studies in late 2021.
M Kamruzzaman. 2024. Bacterial Toxin-Antitoxin Systems. Microorganisms, 12(1):128.
M Kamruzzaman, AJ Mathers, JR Iredell. 2022. A novel plasmid entry exclusion system in pKPC_UVA01, a promiscuous conjugative plasmid carrying the blaKPC carbapenemase gene. Antimicrob. Agents Chemother., 66(3):e02322-21.
M Kamruzzaman and JR Iredell. 2019. CRISPR-Cas system in the antibiotic resistance plasmids in Klebsiella pneumoniae. Front. Microbiol., 10:2934.
Q Qi, M Kamruzzaman, JR Iredell. 2023. A Streamlined approach for fluorescence labelling of low-copy-number plasmids for determination of conjugation frequency by flow cytometry. Microorganisms, 11:878.
M Kamruzzaman, A Yan, GC Escarpulli. 2022. CRISPR-Cas systems in bacteria and archaea. Front. Microbiol., 13:887778.
Kamruzzaman M, Wu AY, Iredell JR. Biological functions of type II toxin-antitoxin systems in bacteria. Microorganisms. 2021 9(6):1276.
Qi Q, Kamruzzaman M, Iredell JR. The higBA-type toxin-antitoxin system in IncC plasmids is a mobilizable ciprofloxacin-inducible system. mSphere. 2021 6(3):e0042421.
Wu AY, Kamruzzaman M, Iredell JR. Specialised functions of two common plasmid mediated toxin-antitoxin systems, ccdAB and pemIK, in Enterobacteriaceae. PLoS One. 2020 15(6):e0230652.
Kamruzzaman M, Iredell J. A ParDE-family toxin antitoxin system in major resistance plasmids of Enterobacteriaceae confers antibiotic and heat tolerance. Sci Rep. 2019 9(1):9872.
Bustamante P, Iredell JR. The roles of HicBA and a novel toxin-antitoxin-like system, TsxAB, in the stability of IncX4 resistance plasmids in Escherichia coli. J Antimicrob Chemother. 2019 74(3):553-556.
Kamruzzaman M, Shoma S, Thomas CM, Partridge SR, Iredell JR. Plasmid interference for curing antibiotic resistance plasmids in vivo. PLoS One. 2017 12(2):e0172913.
Bustamante P, Iredell JR. Carriage of type II toxin-antitoxin systems by the growing group of IncX plasmids. Plasmid. 2017 91:19-27.
Jon is an Infectious Disease Physician and Microbiologist who divides his time between Westmead Hospital in a combined Infectious Diseases and Microbiology Department and his research which is supported by the NHMRC at the University of Sydney.
Jon is an Infectious Disease Physician and Microbiologist who divides his time between Westmead Hospital in a combined Infectious Diseases and Microbiology Department and his research which is supported by the NHMRC at the University of Sydney. His major interests are in critical infection, including the study of bacterial septic shock, and in bacterial genetics and ecology.
Formal affiliations are (1) Conjoint Professor of Medicine and Microbiology, Sydney Medical School The University of Sydney/The Westmead Institute for Medical Research and the Marie Bashir Institute, (2) Senior Staff Specialist, Western Sydney and Westmead Hospital and Deputy Director Microbiology, Pathology West (NSW Pathology) and Institute of Clinical Pathology and Medical Research, (3) Director, Centre for Infectious Diseases and Microbiology at Westmead Institute for Medical Research. List of publications.
Kamal is a Senior Scientist at the Centre for Infectious Diseases and Microbiology, Westmead Institute for Medical Research and a Senior Lecturer (Faculty of Medicine) at the University of Sydney. He worked as a CRE Research Fellow at the University of Sydney from Sep. 2011 to February 2014.
His Major research expertise is in the field of Bacterial genetics, mobile genetic elements (plasmids and bacteriophages) and antibiotic resistance genetics. His research work has been published in major journals such as Nature and PNAS with >2400 citations. His current research focuses on understanding the biology of antibiotic resistance (AbR) plasmids and developing effective therapies to eradicate AbR plasmids. He developed solutions to cure and prevent antibiotic resistance and successfully tested them in mice and started treating humans to eradicate antibiotic resistance. He has been a Chief Investigator of NHMRC and MRFF grants totaling >$3.2 million AUD.
His current endeavour is to form an investible spin-out company to commercialise plasmid therapy products. He has led the “Plasmid Therapy” team to the completion of the national commercialisation programs CSIRO ON Prime-12 and ON Accelerate-8.
He completed his PhD in the field of Molecular Microbiology at Kyoto University Graduate School of Medicine, Japan, in 2008 with a Monbukagakoshu scholarship (Japanese Government scholarship). He worked as an Assistant Scientist and Associate Scientist (2008-2011) at the Molecular Microbiology and Ecology research group at the International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b). His research at ICDDR,B mainly focused on understanding molecular evolution and pathogenic mechanism of enteric bacterial pathogens.
list of publications is available on Medline Research Gate and on Google Scholar citations.
Plasmid Therapy is currently undergoing a first in human trials.
Plasmid therapy is an experimental method that is only available when all other approaches are already optimised and only under the auspices of a special access scheme defined by the Therapeutic Goods Administration or within a clinical trial.
Enquiries from specialist should be directed to our clinical team for discussion.
